✔ 100% Authentic Product
👁️ Currently Viewing 1445
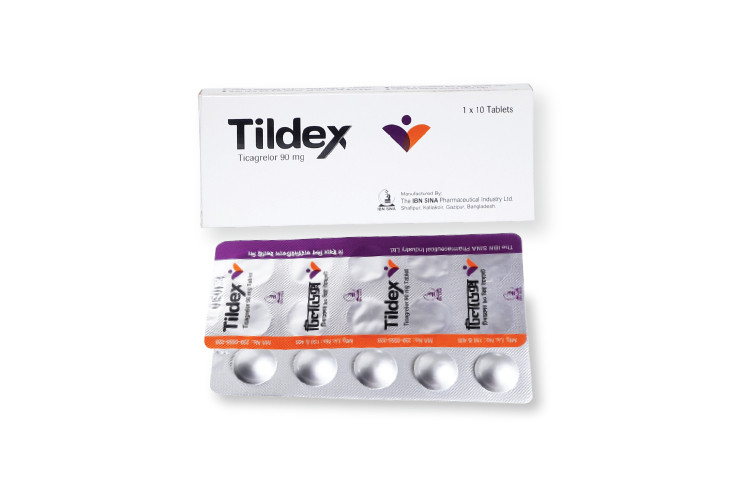
Tildex 60, 1 Strip
Tildex, when used together with acetylsalicylic acid (ASA), is prescribed to help prevent atherothrombotic complications in adults with acute coronary syndrome (ACS) or in patients who have previously experienced myocardial infarction (MI) and remain at high risk for future thrombotic cardiovascular events.
Discount
Price: ৳ 285
MRP:
৳
300
5%
Off

100% Genuine Products, Guaranteed

Safe & Secure Payments, Always

Fast, Secure & Efficient Delivery
Proper Packaging
 Cash on Delivery - All over Bangladesh
Cash on Delivery - All over Bangladesh Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59
Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59 Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125
Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125
 ফ্রি ডেলিভারিঃ - ১৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে
ফ্রি ডেলিভারিঃ - ১৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে ফ্রি ডেলিভারিঃ - ৪৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
ফ্রি ডেলিভারিঃ - ৪৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
📲 মোবাইল অ্যাপ অর্ডারে সাশ্রয় বেশী
-
Google Play Store থেকে ডাউনলোড
-
Apple Store থেকে ডাউনলোড
100% Genuine Products, Guaranteed
Safe & Secure Payments, Always
Fast, Secure & Efficient Delivery
Proper Packaging
 Cash on Delivery - All over Bangladesh
Cash on Delivery - All over Bangladesh Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59
Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59 Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125
Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125 ফ্রি ডেলিভারিঃ - ১৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে
ফ্রি ডেলিভারিঃ - ১৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে ফ্রি ডেলিভারিঃ - ৪৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
ফ্রি ডেলিভারিঃ - ৪৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে- Google Play Store থেকে ডাউনলোড
- Apple Store থেকে ডাউনলোড
📲 মোবাইল অ্যাপ অর্ডারে সাশ্রয় বেশী
✅ Description:
Ticagrelor is an antiplatelet medication that works by selectively blocking the P2Y12 receptor on platelets. This receptor normally contributes to platelet activation and aggregation through signaling pathways involving Gi proteins and related mediators. By inhibiting this receptor, ticagrelor reduces platelet clumping and lowers the formation of blood clots, thereby decreasing the risk of heart attack and ischemic stroke.
Ticagrelor has been tolerated in single doses up to 900 mg. Overdose may lead to gastrointestinal disturbances, shortness of breath, or ventricular pauses. Symptomatic and supportive treatment should be provided.
Relevant Products / প্রাসঙ্গিক পণ্য
Same generic alternatives, ranked by customer interestMore From Ibne Sina Pharmaceuticals / এই ব্র্যান্ডের আরও পণ্য
Popular products from this manufacturer/brand✔️ Dosage & Administration
Patients taking Ticagrelor should generally continue low-dose ASA therapy (75–150 mg daily), unless contraindicated.
Acute Coronary Syndrome (ACS)
- Treatment is initiated with a loading dose of 180 mg (two 90 mg tablets).
- Thereafter, the recommended dose is 90 mg twice daily.
- Therapy is usually continued for 12 months unless discontinuation is medically necessary.
History of Myocardial Infarction
- For extended prevention, the recommended dose is 60 mg twice daily.
- Treatment may continue after completion of the first year of antiplatelet therapy or may be started within 2 years after MI, or within 1 year after stopping previous ADP receptor inhibitor treatment.
- Clinical evidence beyond 3 years of treatment remains limited.
Switching from Other Antiplatelets
- The first dose of ticagrelor should be taken 24 hours after the last dose of the previous antiplatelet medicine.
Missed Dose
- If a dose is missed, the patient should skip the forgotten dose and take the next scheduled dose at the regular time. Double dosing is not recommended.
Special Populations
- Elderly: No dose adjustment required.
- Renal Impairment: No dose adjustment is needed, but use is not recommended in dialysis patients.
- Hepatic Impairment: Contraindicated in severe hepatic impairment; use cautiously in moderate impairment.
- Children: Safety and efficacy in patients below 18 years are not established.
- Ticagrelor may be taken with or without food.
✔️ Effects of Other Medicines on Tildex
- Strong CYP3A4 inhibitors such as clarithromycin, ritonavir, nefazodone, and atazanavir can significantly increase ticagrelor levels and are contraindicated.
- Potent CYP3A inducers like rifampicin may reduce ticagrelor effectiveness and should generally be avoided.
- Caution is advised when used with moderate CYP3A4 or P-gp inhibitors such as verapamil or quinidine.
✔️ Side Effects
Very Common
- Bleeding-related disorders
- Elevated uric acid levels
- Shortness of breath (dyspnoea)
Common
- Headache, dizziness, fainting, vertigo
- Low blood pressure
- Gastrointestinal bleeding, indigestion, nausea, diarrhea, constipation
- Skin rash, itching, bruising, or bleeding under the skin
- Increased creatinine levels
- Bleeding after procedures or trauma
Uncommon
- Allergic reactions, including angioedema
- Confusion
- Intracranial, eye, ear, retroperitoneal, or muscular bleeding
✔️ Effects of Tildex on Other Medicines
- Ticagrelor may increase exposure to medicines metabolized by CYP3A4, including simvastatin and lovastatin; doses above 40 mg are not recommended.
- It may increase levels of atorvastatin slightly, though usually not clinically significant.
- Use caution with medicines having a narrow therapeutic index, including digoxin and cyclosporine.
- Ticagrelor does not significantly affect CYP2C9 substrates such as warfarin or tolbutamide.
- Combined use with SSRIs may raise bleeding risk.
- Care is advised when used with anticoagulants, heparin, enoxaparin, or desmopressin due to additive effects on bleeding.
✔️ Precautions & Warnings
Bleeding Risk
Use carefully in patients with an increased tendency to bleed, including those with recent surgery, trauma, clotting disorders, or gastrointestinal bleeding.
Surgery
Patients should inform healthcare professionals and dentists before undergoing surgery or starting new medications. Ticagrelor should generally be stopped 5 days before elective surgery.
Patients with Prior Stroke
Patients with a history of ischemic stroke may continue therapy for up to 12 months following ACS.
Bradycardia Risk
Use cautiously in individuals prone to slow heart rate or conduction abnormalities.
Respiratory Conditions
Patients with asthma or COPD may have a greater chance of developing dyspnoea during therapy.
Kidney Function & Uric Acid
Ticagrelor may increase serum creatinine and uric acid levels. Renal function should be monitored, especially in elderly patients or those with renal impairment. Caution is advised in patients with gout or hyperuricaemia.
ASA Dose Limitation
High maintenance doses of ASA above 300 mg daily are not recommended when using Tildex.
Premature Discontinuation
Stopping antiplatelet therapy early may increase the risk of cardiovascular death or recurrent myocardial infarction.
✔️ Pregnancy & Lactation
Ticagrelor is generally not recommended during pregnancy. Animal studies suggest that the drug and its metabolites may pass into breast milk. The decision to continue breastfeeding or discontinue therapy should be based on the clinical importance of treatment for the mother and the benefits of breastfeeding for the infant.
✔️ Tildex should not be used in patients with:
- Hypersensitivity to ticagrelor or any component of the formulation
- Active pathological bleeding
- History of intracranial hemorrhage
- Severe liver impairment
✔️ Storage Conditions
Store in a cool, dry place away from light and moisture. Keep out of reach of children.
⚠️Disclaimer:
At ePharma, we’re committed to providing accurate and accessible health information. However, all content is intended for informational purposes only and should not replace medical advice from a qualified physician. Please consult your healthcare provider for personalized guidance. We aim to support, not substitute, the doctor-patient relationship.