
✔ 100% Authentic Product
👁️ Currently Viewing 517
100% Genuine Products, Guaranteed
Safe & Secure Payments, Always
Fast, Secure & Efficient Delivery
Proper Packaging
 Cash on Delivery - All over Bangladesh
Cash on Delivery - All over Bangladesh Regular Delivery - 8-12 Hours, Dhaka City*
Regular Delivery - 8-12 Hours, Dhaka City* Regular Delivery - 24-48 Hours, All Over Bangladesh*
Regular Delivery - 24-48 Hours, All Over Bangladesh* ফ্রি ডেলিভারি! - ১৪৯৯ টাকা+ অর্ডারে ঢাকা
শহরে ।
ফ্রি ডেলিভারি! - ১৪৯৯ টাকা+ অর্ডারে ঢাকা
শহরে । ফ্রি ডেলিভারি! - ২৯৯৯ টাকা+ অর্ডারে ঢাকার
বাহিরে ।
ফ্রি ডেলিভারি! - ২৯৯৯ টাকা+ অর্ডারে ঢাকার
বাহিরে ।
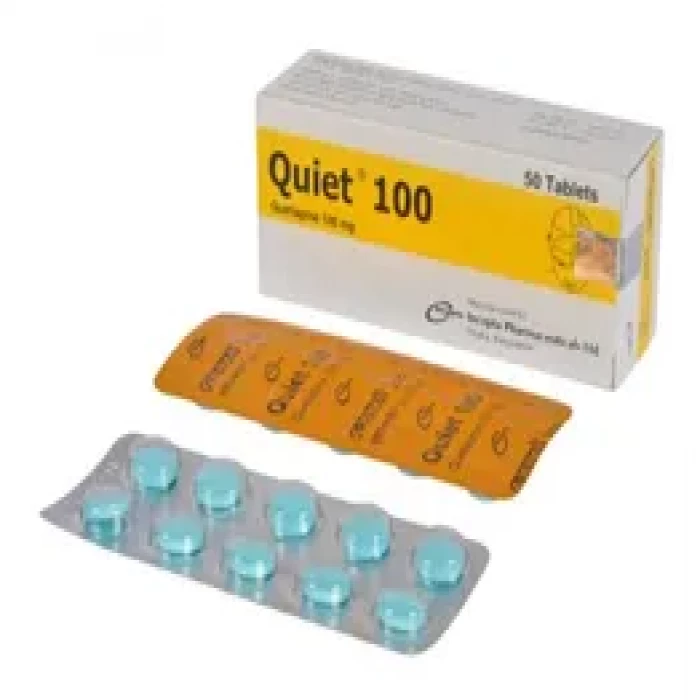
✅ Description:
Quiet 100mg Tablet is used in the treatment of schizophrenia (a mental disorder that can result in hallucinations or delusions and also adversely affects a person’s ability to think and behave) and mania. It is also used to treat bipolar disorders.
Quiet 100mg Tablet may be taken with or without food. However, it is advised to take it at the same time each day as this helps to maintain a consistent level of medicine in the body. Take this in the dose and duration as advised by your doctor and if you have missed a dose, take it as soon as you remember. Quiet 100mg Tablet mustn't be stopped suddenly without talking to your doctor as it may worsen your symptoms. However, discontinue this medicine immediately if you experience Neuroleptic malignant syndrome (NMS), characterized by fever, muscle rigidity, and altered consciousness or seizures.
Safety Advices
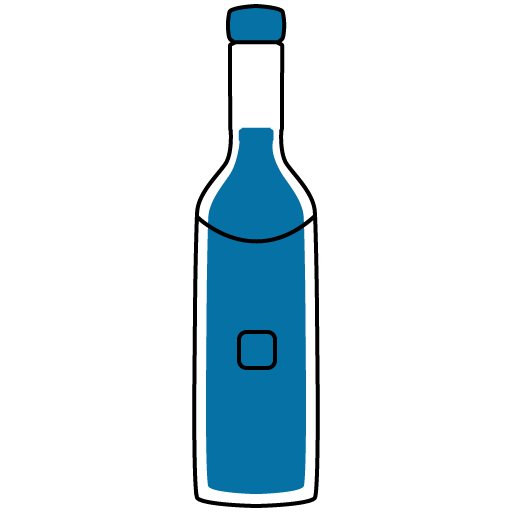
Alcohol
UNSAFE
It is unsafe to consume alcohol while taking Quiet 100mg Tablet. Alcohol can increase the side effects of Qutipin, such as drowsiness, dizziness, and impaired judgment.

Pregnancy
CONSULT YOUR DOCTOR
Quiet 100mg Tablet may be unsafe to use during pregnancy. While there are limited studies in humans, animal studies have shown potential harmful effects on the developing baby. Your doctor will evaluate the benefits and risks before prescribing this medication to you. Please consult your doctor for personalized advice.

CONSULT YOUR DOCTOR
Quiet 100mg Tablet is probably unsafe to use during breastfeeding. Limited human data suggests that the drug may pass into breastmilk and could harm the baby. It is important to consult your doctor before taking this medication if you are breastfeeding.

Driving
CAUTION
Quiet 100mg Tablet may reduce alertness, impair vision, or cause drowsiness and dizziness. Do not drive or operate heavy machinery if you experience these symptoms.

Kidney
SAFE IF PRESCRIBED
Quiet 100mg Tablet is probably safe to use in patients with kidney disease. Limited data suggests that dose adjustment may not be necessary in these patients. However, it is crucial to consult your doctor for personalized advice.

Liver
SAFE IF PRESCRIBED
Quiet 100mg Tablet should be used with caution in patients with liver disease. Dose adjustment may be needed for these patients. Please consult your doctor to determine the appropriate dosage and to monitor liver function during treatment.
✔️ Uses:
- Schizophrenia
- Mania
✔️ How does
Quetiapine is an atypical antipsychotic. It works by modulating the action of certain chemical messengers in the brain that affect thoughts.
✔️ Side Effects:
Common adverse effects include somnolence, dizziness, dry mouth, withdrawal symptoms, elevated serum triglycerides, elevated LDL cholesterol, decreased HDL cholesterol, weight gain, decreased hemoglobin, and extrapyramidal symptoms.
✔️ Quick Suggestions:
- It is less likely to cause heart problems and movement disorders than other antipsychotics.
- It may take 4 to 6 weeks to see treatment effects. Keep taking it as prescribed.
- Use caution while driving or doing anything that requires concentration as Quetiapine can cause dizziness and sleepiness.
- To lower the chance of feeling dizzy or passing out, rise slowly if you have been sitting or lying down.
- It may increase your weight, blood sugar, and cholesterol. Eat healthy, exercise regularly, and monitor your blood levels regularly.
- Inform your doctor if you develop any unusual changes in mood or behavior, new or worsening depression, or suicidal thoughts or behavior.
- Do not stop taking it without talking to your doctor first as it may cause worsening of symptoms.
✔️ Indications:
Quiet is indicated for the treatment of acute and chronic psychoses, including schizophrenia, and bipolar disorder. This includes the treatment of manic episodes meeting DSM-IV criteria for mania, treatment of depressive episodes associated with bipolar disorder, and maintenance treatment of bipolar I disorder in combination with a mood stabilizer to prevent the recurrence of manic, depressive, or mixed episodes.
✔️ Pharmacology
Quetiapine Fumarate is an atypical antipsychotic from the dibenzothiazepine derivative class. It antagonizes multiple neurotransmitter receptors in the brain, including serotonin 5HT1A and 5HT2, dopamine D1 and D2, histamine H1, and adrenergic α1 and α2 receptors. It does not significantly bind to cholinergic muscarinic and benzodiazepine receptors. The efficacy in schizophrenia is thought to be due to dopamine D2 and serotonin 5HT2 antagonism. Quetiapine's antagonism of histamine H1 receptors can cause somnolence, and its effect on adrenergic receptors can lead to orthostatic hypotension.
✔️ Elderly:
Use with caution, start with lower doses, and adjust slowly based on response and tolerability.
✔️ Overdose Effects
In cases of overdose, establish and maintain a patent airway, ensure adequate oxygenation and ventilation, and monitor cardiovascular function. Treat refractory hypotension with intravenous fluids and/or sympathomimetic agents, avoiding beta stimulants like adrenaline and dopamine.
✔️ Children and Adolescents:
Not indicated for use in individuals below 18 years of age.
✔️ Dosage & Administration:
- Acute and Chronic Psychoses (including Schizophrenia): Administer twice daily with or without food. Start with 50 mg on Day 1, increase to 100 mg on Day 2, 200 mg on Day 3, and 300 mg on Day 4. Adjust dosage from Day 4 onwards to an effective range of 300-450 mg/day, with possible adjustments between 150 and 750 mg/day based on clinical response and tolerability.
- Manic Episodes (Bipolar Disorder): Administer twice daily with or without food. Start with 100 mg on Day 1, increase to 200 mg on Day 2, 300 mg on Day 3, and 400 mg on Day 4. Dosage can be adjusted up to 800 mg/day by Day 6 in increments of no more than 200 mg/day. The effective dose range is 400-800 mg/day.
- Depressive Episodes (Bipolar Disorder): Administer once daily at bedtime with or without food. Start with 50 mg on Day 1, increase to 100 mg on Day 2, 200 mg on Day 3, and 300 mg on Day 4. Dosage can be increased to 400 mg on Day 5 and up to 600 mg by Day 8. Efficacy was demonstrated at both 300 mg and 600 mg doses.
- Maintenance Treatment (Bipolar I Disorder): Continue the effective dose from the acute phase, adjusting as necessary based on clinical response and tolerability. Effective doses range from 400 to 800 mg/day.
✔️ Interaction
- Electrolyte Imbalance & QT Prolongation: Use caution with medications causing these effects.
- Increased Clearance: Co-administration with thioridazine, carbamazepine, or phenytoin increases quetiapine clearance.
- Contraindications: Hypersensitivity to quetiapine.
✔️ Contraindications
Hypersensitivity
✔️ Pregnancy & Lactation
Quetiapine should only be used during pregnancy if the benefits outweigh the potential risks, and the dose should be kept as low and as short as possible. The excretion of quetiapine in human milk is unknown; therefore, breastfeeding while taking quetiapine is not recommended.
✔️ Precautions & Warnings
- Suicidal Thoughts: Depression increases the risk of suicide-related events until significant remission.
- Cardiovascular Disease: Use with caution in patients with cardiovascular or cerebrovascular disease or conditions predisposing to hypotension.
- Seizures: Use caution in patients with a history of seizures.
- Tardive Dyskinesia & EPS: Consider dose reduction or discontinuation if symptoms appear.
- Neuroleptic Malignant Syndrome: Discontinue and provide appropriate treatment if symptoms occur.
- QT Prolongation: Exercise caution in patients with cardiovascular disease or family history of QT prolongation.
- Neutropenia: Monitor patients for severe neutropenia, especially in the first two months.
- Hyperglycemia & Diabetes: Monitor for increases in blood glucose levels and symptoms of diabetes.
✔️ Storage Conditions:
Store in a cool, dry place away from light. Keep out of reach of children. Do not use after the expiry date stated on the packaging.
Disclaimer:
ePharma sole intention is to ensure that its consumers get proper
information as musch as possible. Although we do not guarantee the
accuracy and the completeness of the information that provided and
here information is for informational purposes only.
The information contained herein should NOT be used as a substitute
for the advice of a qualified physician. This may not cover
everything about particular health conditions,
lab tests, medicines, all possible side effects, drug interactions,
warnings, alerts, etc. Please consult your healthcare professional
and discuss all your queries related to any disease or medicine. We
intend to support, not replace, the doctor-patient relationship.