✔ 100% Authentic Product
👁️ Currently Viewing 3089
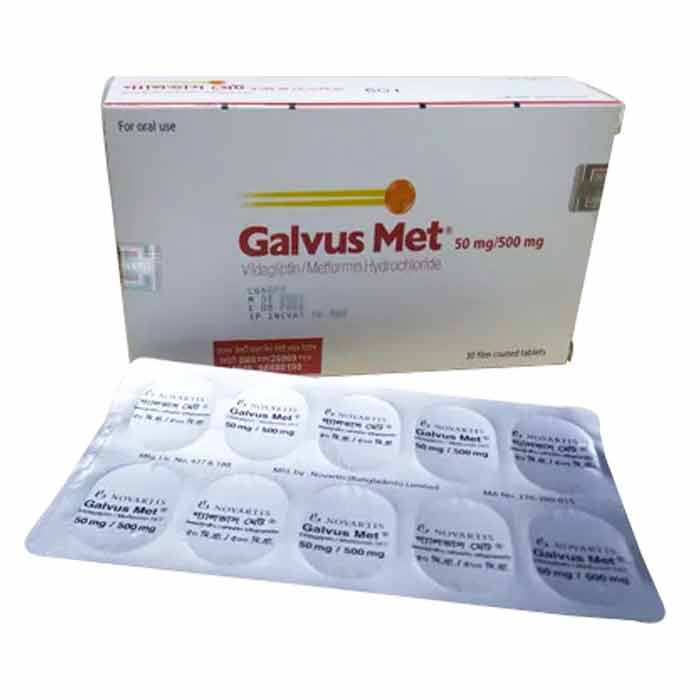
Galvus Met 50/500mg 10pcs
Tablet. Galvus Met 50mg/500mg Tablet is used in the treatment of type 2 diabetes.
Discount
Price: ৳ 320
MRP:
৳
330
3%
Off

100% Genuine Products, Guaranteed

Safe & Secure Payments, Always

Fast, Secure & Efficient Delivery
Proper Packaging
 Cash on Delivery - All over Bangladesh
Cash on Delivery - All over Bangladesh Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59
Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59 Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125
Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125
 ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে
ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে ফ্রি ডেলিভারিঃ - ৫৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
ফ্রি ডেলিভারিঃ - ৫৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
📲 মোবাইল অ্যাপ অর্ডারে সাশ্রয় বেশী
-
Google Play Store থেকে ডাউনলোড
-
Apple Store থেকে ডাউনলোড
100% Genuine Products, Guaranteed
Safe & Secure Payments, Always
Fast, Secure & Efficient Delivery
Proper Packaging
 Cash on Delivery - All over Bangladesh
Cash on Delivery - All over Bangladesh Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59
Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59 Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125
Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125 ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে
ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে ফ্রি ডেলিভারিঃ - ৫৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
ফ্রি ডেলিভারিঃ - ৫৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে- Google Play Store থেকে ডাউনলোড
- Apple Store থেকে ডাউনলোড
📲 মোবাইল অ্যাপ অর্ডারে সাশ্রয় বেশী
✅ Description:
Galvus Met 50mg/500mg Tablet is a combination of two antidiabetic medicines: Metformin and vildagliptin. Metformin is a biguanide which decreases the production and absorption of sugar in your body and allows better use of existing insulin. Vildagliptin is a dipeptidyl peptidase-4 inhibitor which works by increasing the release of insulin from pancreas and decreasing the hormones that raise blood sugar levels. This reduces both fasting and postmeal sugar levels. Together, they provide better control of blood sugar.
Dosage & Administration of Galvus Met 50mg/500mg
Adults: Based on the patient's current dose of Metformin, this combination may be initiated twice daily, with 1 tablet in the morning and the other in the evening. Patients receiving Vildagliptin and Metformin from separate tablets may be switched to this combination containing the same doses of each component. Doses higher than 100 mg of vildagliptin are not recommended. There is no clinical experience of Vildagliptin and Metformin in triple combination with other antidiabetic agents. Taking this combination with or just after food may reduce gastrointestinal symptoms associated with Metformin.
Interaction of Galvus Met 50mg/500mg
No clinically relevant pharmacokinetic interaction was observed when Vildagliptin (100 mg once daily) was co-administered with Metformin Hydrochloride (1,000 mg once daily). Vildagliptin has a low potential for drug interactions. Since Vildagliptin is not a cytochrome P (CYP) 450 enzyme-substrate nor does it inhibit nor induce CYP 450 enzymes, it is not likely to interact with co-medications that are substrates, inhibitors or inducers of these enzymes. As a result of these studies no clinically relevant interactions with other oral antidiabetics (glibenclamide, pioglitazone, metformin hydrochloride), amlodipine, digoxin, ramipril, simvastatin, valsartan or warfarin were observed after co-administration with vildagliptin. On the other hand, furosemide, nifedipine and glyburide increase Cmax and blood AUC of Metformin with no change in renal clearance of Metformin.
Contraindications
This combination is contraindicated in patients with known hypersensitivity to Vildagliptin or Metformin Hydrochloride or to any of the excipients. It is contraindicated in patients with renal disease or renal dysfunction, acute myocardial infarction, and septicaemia. It is also contraindicated in patients with congestive heart failure patients and in patients with acute or chronic metabolic acidosis, including diabetic ketoacidosis, with or without coma. It should be temporarily discontinued in patients undergoing radiologic studies involving intravascular administration of iodinated contrast materials, because the use of such products may result in acute alteration of renal function.
Side Effects of Galvus Met 50mg/500mg
The most common side effects are headache, tremor, dizziness, nausea, hypoglycaemia etc.
Pregnancy & Lactation while using Galvus Met 50mg/500mg
There are no adequate and well-controlled studies in pregnant women and therefore, this combination should not be used during pregnancy unless the potential benefit justifies the potential risk to the foetus. No studies have been conducted on the components of this combination. As it is not known whether Vildagliptin and/or Metformin Hydrochloride is excreted in human milk this combination should not be administered to breastfeeding women.
Precautions & Warnings of Galvus Met 50mg/500mg
Lactic acidosis can occur due to Metformin accumulation. If metabolic acidosis is suspected, treatment should be discontinued and the patient should be hospitalized immediately. Serum creatinine should be monitored at least once a year in patients with normal renal function and 2–4 times a year in patients with serum creatinine levels at the upper limit of normal and in elderly patients. Special caution should be exercised in elderly patients where renal function may become impaired (e.g. when initiating antihypertensives, diuretics or NSAIDs). It is recommended that Liver Function Tests (LFTs) are monitored prior to initiation of this drug, at three-monthly intervals in the first year and periodically thereafter. If transaminase levels are increased, patients should be monitored with a second liver function evaluation to confirm the finding and be followed thereafter with frequent liver function tests until the abnormality return to normal. If AST or ALT persist at 3 x ULN, Vildagliptin & Metformin tablets should be stopped for Patients who develop jaundice or other signs of liver dysfunction. Following the withdrawal of treatment with Vildagliptin & Metformin and LFT normalization, treatment with Vildagliptin & Metformin should not be reinitiated. Vildagliptin & Metformin tablets should be discontinued 48 hours before elective surgery with general anaesthesia and should not usually be resumed earlier than 48 hours afterwards.
⚠️Disclaimer:
At ePharma, we’re committed to providing accurate and accessible health information. However, all content is intended for informational purposes only and should not replace medical advice from a qualified physician. Please consult your healthcare provider for personalized guidance. We aim to support, not substitute, the doctor-patient relationship.