✔ 100% Authentic Product
👁️ Currently Viewing 2808
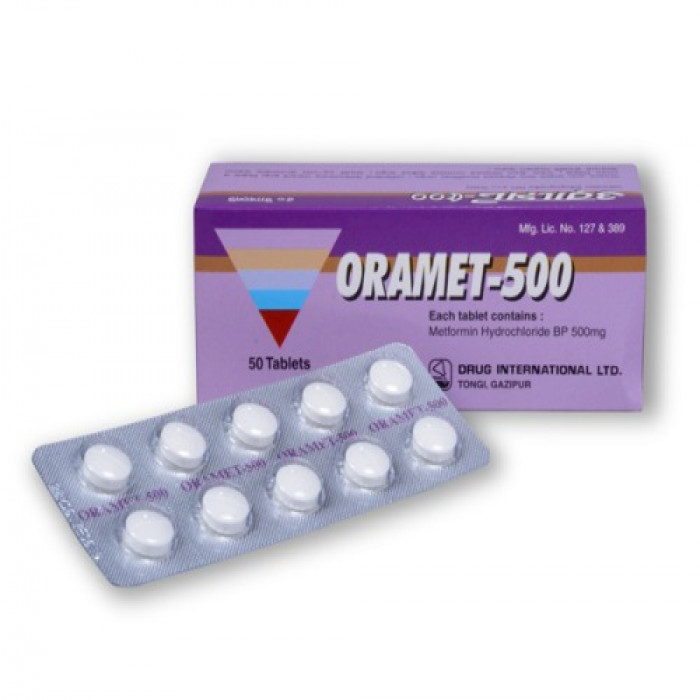
Oramet 500mg 10pcs
Tablet. Manufacturer/Distributor: Drug International Ltd. Generic Name: Metformin Hydrochloride 500 mg
Metformin Hydrochloride, as monotherapy, is indicated as an adjunct to diet to lower blood glucose especially in overweight patients with non-insulin-dependent diabetes mellitus (NIDDM) or type 2 diabetes mellitus whose hyperglycemia cannot be satisfactorily managed on diet alone.
Discount
Price: ৳ 38
MRP:
৳
40
5%
Off

100% Genuine Products, Guaranteed

Safe & Secure Payments, Always

Fast, Secure & Efficient Delivery
Proper Packaging
 Cash on Delivery - All over Bangladesh
Cash on Delivery - All over Bangladesh Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59
Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59 Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125
Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125
 ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে
ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে ফ্রি ডেলিভারিঃ - ৫৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
ফ্রি ডেলিভারিঃ - ৫৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
📲 মোবাইল অ্যাপ অর্ডারে সাশ্রয় বেশী
-
Google Play Store থেকে ডাউনলোড
-
Apple Store থেকে ডাউনলোড
100% Genuine Products, Guaranteed
Safe & Secure Payments, Always
Fast, Secure & Efficient Delivery
Proper Packaging
 Cash on Delivery - All over Bangladesh
Cash on Delivery - All over Bangladesh Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59
Regular Delivery: 24-48 Hours, Dhaka City* Charge Tk.39-59 Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125
Regular Delivery: 48-96 Hours, Other Cities* Charge Tk.99-125 ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে
ফ্রি ডেলিভারিঃ - ২৯৯৯ টাকা+ অর্ডারে, ঢাকা
শহরে ফ্রি ডেলিভারিঃ - ৫৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে
ফ্রি ডেলিভারিঃ - ৫৯৯৯ টাকা+ অর্ডারে, ঢাকার
বাহিরে- Google Play Store থেকে ডাউনলোড
- Apple Store থেকে ডাউনলোড
📲 মোবাইল অ্যাপ অর্ডারে সাশ্রয় বেশী
✅ Description:
Metformin immediate release tablet: Dosage of Metformin Hydrochloride must be individualized on the basis of both effectiveness and tolerance, while not exceeding the maximum recommended daily doses.
- Adult: The usual starting dose of Metformin is 500 mg twice a day or 850 mg once a day, given with meals. Dosage increases should be made in increments of 500 mg weekly or 850 mg every 2 weeks, up to a total of 2000 mg per day, given in divided doses. For those patients requiring additional glycemic control, Glucomin may be given to a maximum daily dose of 2550 mg per day. Doses above 2000 mg may be better tolerated given three times a day with meals.
- Children: The usual starting dose of Metformin is 500 mg twice a day, given with meals. Dosage increases should be made in increments of 500 mg weekly up to a maximum of 2000 mg per day, given in divided doses.
Metformin extended release tablet: Swallow Metformin XR tablet whole and never crush, cut or chew.
- Adult: The usual starting dose of Metformin XR is 500 mg once daily with the evening meal. Dose should be increased in increments of 500 mg weekly, up to a maximum of 2000 mg once daily with the evening meal, alternatively increased to 1000 mg twice daily taken with meal. Patient receiving Metformin immediate release tablet may be switched to Metformin extended release tablet up to a maximum recommended daily dose.
- Children: Metformin extended release tablet has not been studied in children.
- Renal impaired patient: Do not use Metformin in patients with eGFR below 30 mL/min/1.73 m2. Asses risk/benefit of counting if eGFR falls below 45 mL/min/1.73 m2.
⚠️Disclaimer:
At ePharma, we’re committed to providing accurate and accessible health information. However, all content is intended for informational purposes only and should not replace medical advice from a qualified physician. Please consult your healthcare provider for personalized guidance. We aim to support, not substitute, the doctor-patient relationship.